|
The construction of a Knudsen double cell-mass spectrometer apparatus designed to measure caesium-graphite equilibrium vapour pressures is also described. Evidence for sensitivity of diffusion coefficients to graphite structure and thermal history was found, although no evidence for the dependence of diffusion coefficients upon the concentration of caesium was found. Caesium is one of four elements who exist in liquid form at or near room temperature. Desorption from the fine grain graphite was controlled by mixed diffusion. Caesium is a silvery-gold metallic element. Place the following elements in order of decreasing atomic size. Each element in the periodic table has a distinctive atomic radius. Slow diffusion in gilsonite graphite was described by log(D) = -5.1 - 137000/2.303 RT. potassium Which element has the smaller atomic radius: Cl or F Explain. 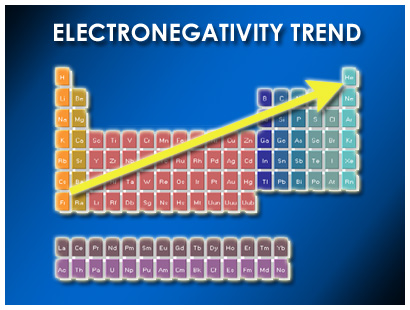
This behaviour was qualitatively explained in terms of a two part diffusion mechanism combined with surface evaporation. The concentration profiles for caesium in graphite, found by electron probe microanalysis, showed evidence of a complex mechanism for transport of caesium in graphite with apparent violations of Fick's Law. It forms alloys with alkali metals, gold and mercury. It is outstanding in keeping time with precision, so it is used in atomic clocks. The effects of a non-uniform concentration of caesium and a surface evaporation step on the transport of caesium in the graphite were also considered. Caesium, also spelt as Cesium, was discovered in 1860. Cesium (spelled 'caesium' internationally, but 'cesium' in the U.S.) is the metallic chemical element represented by the symbol Cs on the periodic table of elements. length of the Paris metre and the duration of the day, but that has no significance. The desorption curves were analysed to obtain diffusion coefficients by several techniques and the validity of each method is considered. We would then have found that the caesium period (T) was changing. 
One of the worlds richest sources of cesium is located at Bernic Lake, Manitoba. Cesium, an alkali metal, occurs in lepidolite, pollucte (a hydrated silicate of aluminum and cesium), and in other sources. The desorption curves consisted of an initial fast period controlled by evaporation of caesium from the surface, followed by a slow rate, controlled by diffusion from the bulk of the graphite. Cesium was discovered spectroscopically in 1860 by Bunsen and Kirchhoff in mineral water from Durkheim. Concentration profiles have been obtained at various stages in sin adsorption, heat treatment, desorption programme by electron probe microanalysis. AbstractThe sorption and diffusion of caesium in two nuclear graphites at a concentration range of 5 x 10-2 to 10-3 atom % and at temperatures in the range 500 to 1400 has been studied using a microgravimetric desorption technique.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
